 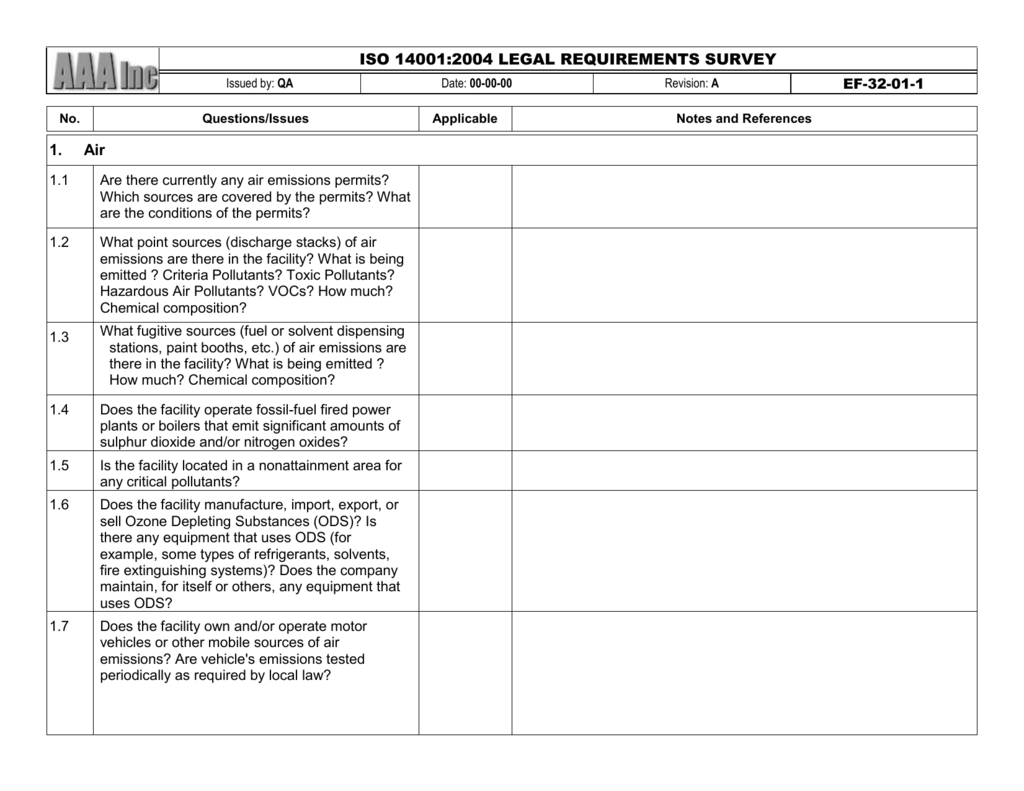
Use this ISO 13485 checklist to determine whether your company’s QMS meets the ISO standards and take corrective actions. A QMS will typically include processes and procedures for analyzing the quality of products and services, managing risk related to quality issues, maintaining records of performance (including corrective actions), and training employees on how to use the system. An ISO13485 audit checklist can be used as part of this process. To prepare for an ISO 13487 auditing process, it is important to have baseline information about your organization’s QMs already in place. This provides assurance that your organization meets global safety and quality standards so that you can maintain customers’ trust and protect your business reputation. Note: For the exam portion of the course, each attendee is required to. Our Medical Device Quality Assurance and Regulatory Affairs document templates have been created by fully qualified and experienced professionals at Patient Guard, a Quality Assurance and Regulatory Affairs Service provider. ISO 13485 talks about two main components of internal audits (section 8.2. This three-day class begins with foundational knowledge of the ISO 13485 standard and builds upon it to prepare you for real-life auditing situationsincluding managing the audit process and completing reportingvia hands-on workshops and two competency exams (MD & AU). An ISO 13485 audit checklist is utilized by quality managers to determine if the organization’s QMS is aligned with the ISO 13485:2016 standard. 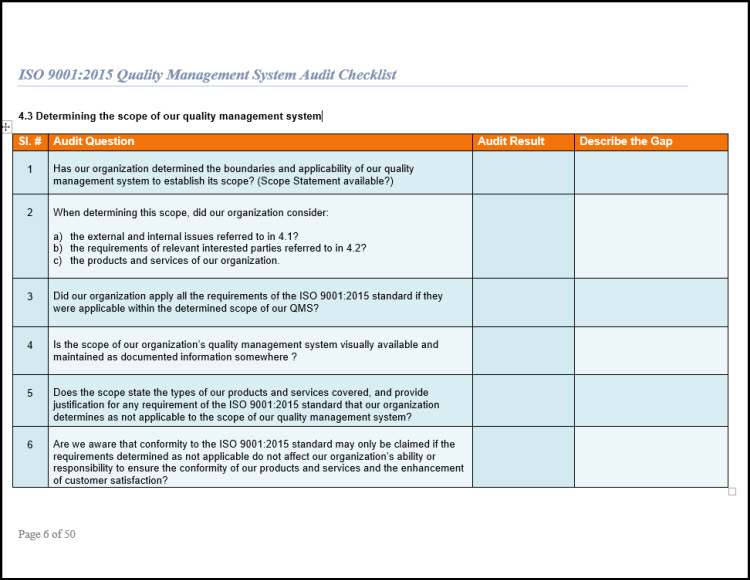
A certification audit involves conducting an in-depth evaluation of a company’s QMS to determine its compliance with specific standards. ISO 13485: 2016 +A11 2021 QMS.8.2.4.2 - QR - Internal Audit Plan & Checklist Template. Internal Audit Checklist ISO 13485 templates ISO 13485 / MDR document template: Internal Audit Checklist. ISO 13485 requirements checklists help assess whether a company is ready to undergo an ISO 13485 certification audit by a third party. The ISO 13485:2016 standard establishes a framework for the quality management system (QMS) requirements for healthcare products and services. Then in columns B -> list the ISO 13485 clauses that these processes will cover. Purchasing, Design, Quality Control, Equipment Maintenance and Calibration). It’s important for businesses to be compliant with these standards because it shows they are taking their quality management seriously. ALEONARD said: Make it simple on yourself, use Excel and list your core business processes in column A (e.g. ISO 13485 is a set of international standards that help organizations identify, control, and prevent quality problems. Inspect your quality management system with an ISO 13485 audit checklist 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
